Your best hair & skin, delivered
From the online visit to contact-free delivery with ongoing care, expect a seamless derm experience.
Dermatologist-tested
Personalized treatment
Confidence in your hair and skin


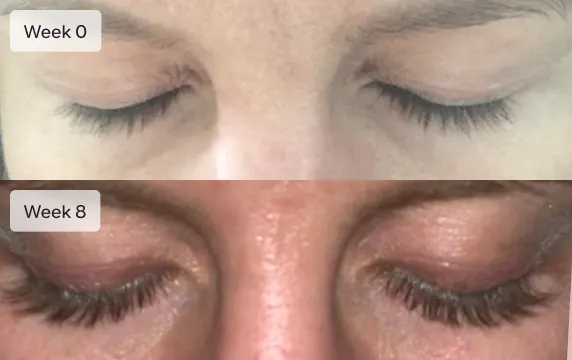
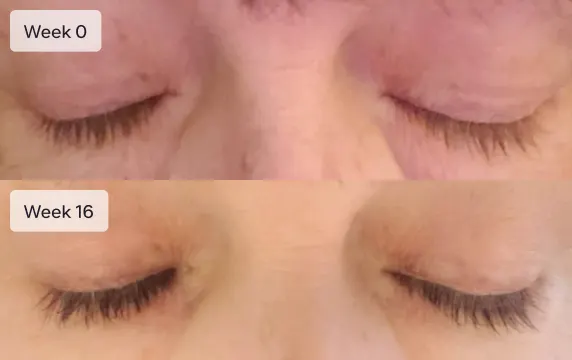
Longer lashes
Achieve a more youthful, revitalized look through prescription eye drops that temporarily lift drooping eyelids.


Fuller hair
Ro offers prescription medication, topical treatment, and over-the-counter products to help you meet your hair goals.


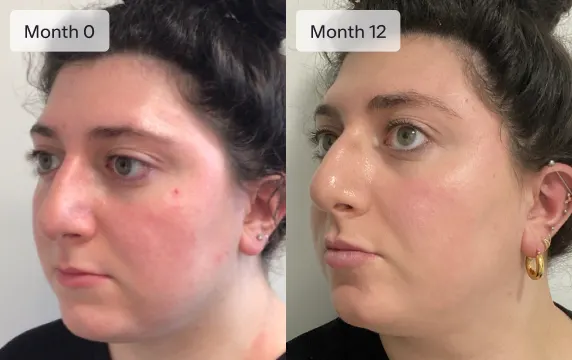
Healthier skin
Ro provides dermatologist-backed offerings from the comfort of your phone. If appropriate, get prescription treatments for your skin to help you reach your goals.
Our derm philosophy
Ro is backed by science and expertise. Our dermatologist-designed experience empowers people to feel their most confident. We developed our offerings with your goals in mind, meaning we selected products and ingredients based on medical knowledge and experience.
We acknowledge that “clean beauty” is an unregulated term, but we understand that people have preferences about the products they use. With guidance from our in-house dermatologists, we have decided not to formulate with the most common skin allergens, per the American Contact Dermatitis Society.
We’re here to help you understand your timeline for results, so we’re clear about what to expect and when to expect it. “Your best skin” is different for everyone—we all have pores, pimples, lines, or wrinkles—so we celebrate your present as well as your future. Our emphasis is on healthy skin, so we steer clear of words like “flawless” or “poreless” to set realistic expectations.
We get it—aging is a gift and we don’t take it for granted. But, at the same time, we believe that people have a right to age in a way that makes them feel good. Our goal is to remove the stigma that comes with aging, as well as the stigma that surrounds seeking anti-aging treatment.
Acne happens, and it hurts, both emotionally and physically. Our goal is to make sure you feel good and feel seen throughout your skincare journey, no matter your starting point. That’s why you see images of real skin at all stages—because the before, the during, and the after are all beautiful.
Join 250,000+ Ro patients
Hear more from our verified members
"I have lighter hair and I was so impressed with how thick and dark my lashes grew in."
"My results have been so much better than I dared hope."
"I was spending so much on OTC skincare and not getting the results I wanted, and I decided to try Ro to get scrip strength without all the visits and hassle."
We don’t want to brag... So we’ll let our press coverage do it for us*
*These press highlights were originally written about Rory and Nightly Defense skincare treatment, which have since been renamed as Ro and Custom Rx Treatment, respectively. The treatment formulations did not change when the product was renamed.
"One of "Allure Editors' Favorite New Beauty Products" in April 2021"
"A digital haven for health and beauty concerns"
"Sends your custom treatment via two-day shipping in sleek packaging, so you can start your new routine almost immediately"
"Combats acne, texture, and fine lines with a...customized blend of tretinoin, niacinamide, ceramides, and vitamin E"
"Virtually connects you to medical professionals; I was still able to get access to the prescription-grade stuff, which they custom blend"
"A month later, my skin has been looking radiant and healthy even though I've been barely getting any sun"
"The new standard of online skincare and telehealth"
FAQ
Ro uses telemedicine technology to connect you with a US-licensed healthcare professional to provide you with prescription treatment to address your skin concerns conveniently and affordably.
It starts with your online visit. The healthcare professional needs to know about your general health and your skin concerns. If required, they may need an unedited photo of you and your ID (that shows your picture and birthdate), so they know who they will be helping. For certain conditions, a photo of the affected area is typically also needed to evaluate your skin.
The healthcare professional reviews everything, determines if you’re a candidate for telemedicine and whether treatment is right for you, and, if so, prescribes the appropriate treatment.
Along with your prescription, your doctor or nurse practitioner will provide you with a treatment plan that includes a great deal of information about your treatment as well as other useful health information. It’s important that you take the time to read it all to be informed and better prepared to take care of your skin.
Not only will your provider start you off with prescription treatment, but they will partner with you on your healthcare journey, and, as your skin evolves, they will re-evaluate and, if appropriate, make adjustments to your treatment.
Latisse is an FDA-approved treatment to grow eyelashes for people with inadequate or not enough lashes. Latisse is clinically-proven to make eyelashes longer, thicker, and darker. The active ingredient in Latisse is bimatoprost ophthalmic solution 0.03%.
Upneeq® (oxymetazoline hydrochloride ophthalmic solution), 0.1% is a prescription eye drop used to treat acquired ptosis in adults.
Acquired ptosis, also known as low-lying lids, is a common condition that occurs when the muscles in the eyelid stretch and weaken, causing the top eyelid to drop.
Acquired ptosis affects adults of all ages, but occurs more often with increased age. Acquired ptosis can also be a result of cataract surgery, contact lens wear, or an underlying medical condition. It’s not uncommon for patients to have acquired ptosis in only one eye or in one eye more than the other.
Custom Rx Treatment is made with a blend of ingredients that were hand-selected by a dermatologist and customized to fit your needs. Depending on your specific skin type, concerns, and goals, the ingredients in your Custom Rx Treatment may include:
Tretinoin (see Important Safety Information)
Azelaic acid (see Important Safety Information)
Niacinamide
Vitamin E acetate
Tranexamic acid
Ceramides
Hyaluronic acid
If prescribed, you will receive a detailed treatment plan that describes which ingredients are included in your custom formulation.